Plasma medicine

Introduction
Research conducted at LPP in the field of “plasma medicine” focuses on the development and characterization of new low-temperature plasma sources. The main objective of our work is to provide advanced therapeutic options for wound healing and oncology, particularly in cases where standard treatment methods prove limited.
The therapeutic efficacy of our cold plasma sources is demonstrated through collaborations with biologists and clinicians, with the ultimate goal of enabling technology transfer to hospital settings.
Medical applications studied at the LPP
Skin wound healing
A major challenge is improving the management of complex skin wounds often involving tissue damage and vascular disorders. Within a large collaborative program with the French Armed Forces Biomedical Research Institute, Institut Pasteur, BIOC (École Polytechnique), and CTI Biotech, we have demonstrated that cold plasmas activate four fundamental pillars of skin wound healing:
i) stimulation of epidermal and dermal cell growth,
ii) stimulation of extracellular matrix protein production (dermis),
iii) stimulation of angiogenesis,
iv) direct bactericidal effects and indirect antibacterial action via immune cell activation.
Cold plasma treatments are designed to be integrated into existing care protocols through applications lasting a few tens of seconds during dressing changes. The treatment is painless, as only the cold plasma jet comes into contact with the wound.
In a preclinical model of severe burns, the treatment accelerates graft acceptance, even under septic conditions. In a preclinical model of chronic wounds, the treatment reduces healing time by 50% and lowers the risk of infection.
This program is supported by the French Directorate General of Armaments, the Defense Innovation Agency, École Polytechnique, Institut Polytechnique de Paris, the École Polytechnique Foundation, SATT Saclay, and Plasana Medical.
The startup Plasana Medical was founded in 2022 to develop a medical device dedicated to wound healing.
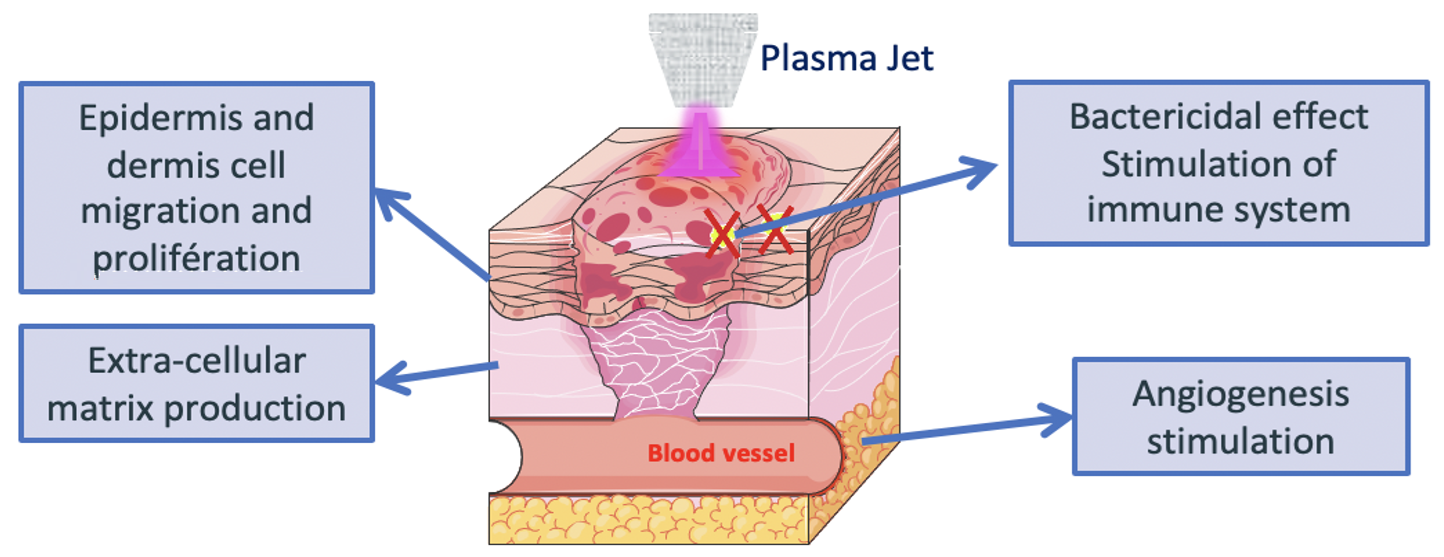
Figure 1 : Cold plasma triggers 4 pillars of wound healing via Reactive Oxygen Species and Electric field
Cancerology
In oncology, the laboratory focuses on the treatment of two poor-prognosis cancers:
Cholangiocarcinoma (bile duct tumors), studied in collaboration with Dr. Laura Fouassier (NABI Laboratory – Nanomedicine, Extracellular Biology, Integratome and Health Innovations).
Non-small cell lung cancer (NSCLC), investigated in partnership with Prof. Isabelle Cremer (Centre de Recherche des Cordeliers, CRC).
The properties of cold plasma are studied to induce and control several biological effects:
Selective targeting of cancer cells through oxidative stress leading to apoptosis, while preserving surrounding healthy tissues;
Remodeling of the extracellular matrix of “cold” tumors, for example by promoting immune cell infiltration, including dendritic cells, macrophages, and B lymphocytes.
This approach opens new perspectives for localized cancer treatment, either as an alternative or complementary strategy to conventional methods such as surgery, chemotherapy, and radiotherapy.
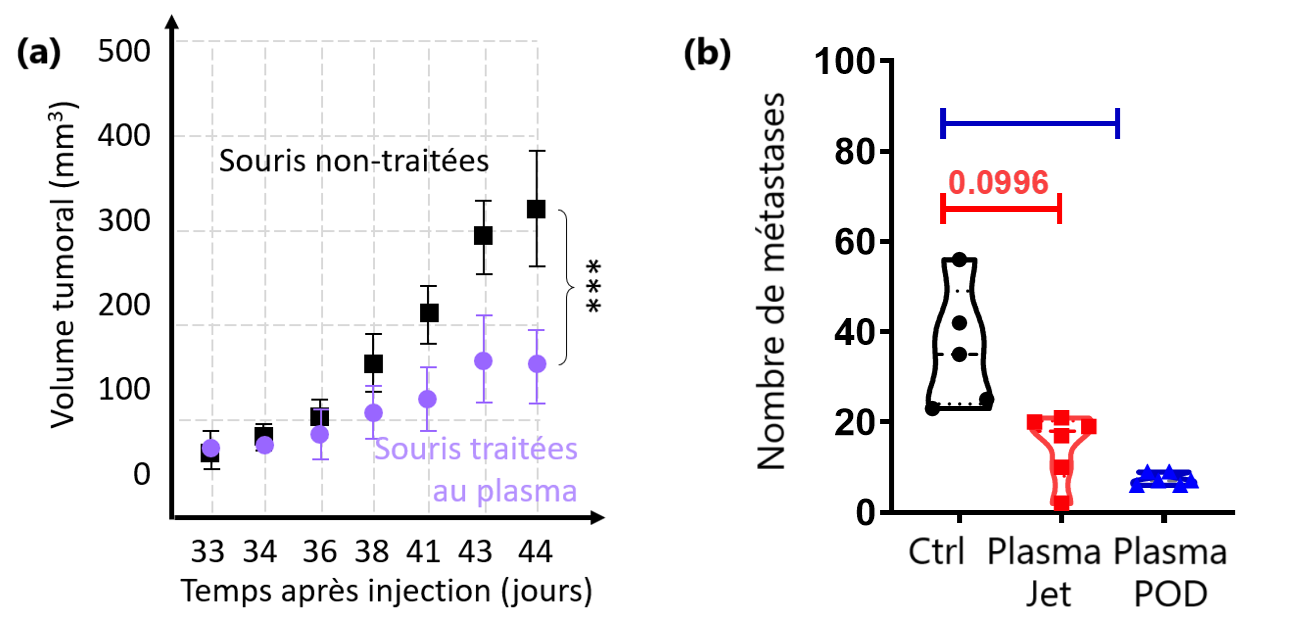
Figure 2. (a) Influence of cold plasma on tumor volume in immunocompetent mice bearing subcutaneous cholangiocarcinoma grafts (NABI–LPP collaboration). (b) Influence of two cold plasma sources (plasma jet and POD plasma) on the number of metastases counted in murine models of non-small cell lung cancer (CRC–LPP collaboration).
Plasma sources dedicated to medicine
The biological and therapeutic effects described above would not have been possible without prior engineering and characterization of the plasma sources. Regardless of the targeted pathology, each source must meet stringent specifications combining:
A “Feasibility” component (e.g., electrical safety, temperature below 40°C),
A “Therapeutic efficacy” component (i.e., identification and optimization of cold plasma properties responsible for the desired therapeutic effects).
The cold plasma sources developed at the LPP for medical applications are plasma jets. The basic principle consists of a tubular dielectric device supplied with gas and electrical power to generate a plasma plume (5–30 mm in length). This technological building block can then be adapted to meet the specific requirements of medical applications.
For the treatment of skin wounds such as severe burns and chronic wounds, we have developed a multi-jet medical device dedicated to rapid wound treatment during dressing changes.
For internal pathologies such as solid cholangiocarcinoma tumors, we have developed a series of cold plasma catheters compatible with endoscopic tools routinely used in hospitals. These catheters can be inserted into the working channel of conventional duodenoscopes to deliver cold plasma doses in situ.
These sources are tested and validated by clinicians (Surgical School, Saint-Antoine Hospital, French Armed Forces Biomedical Research Institute, Pasteur Institute, and external laboratories) on preclinical models. A multicenter clinical trial is currently underway in the field of chronic wound healing.
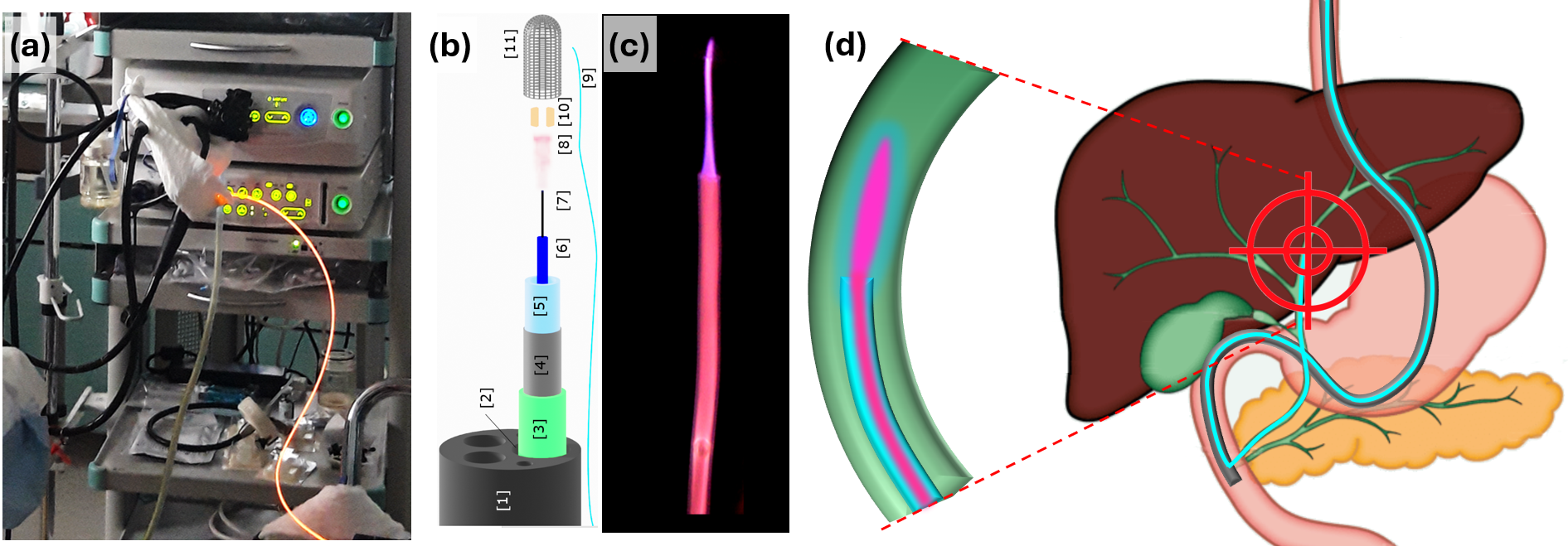
Figure 3. (a) Cold plasma endoscopy column with plasma transferred into a flexible catheter; (b) exploded view of a cold plasma catheter inserted into a duodenoscope; (c) photograph of the distal end of a cold plasma catheter; (d) schematic representation of catheter insertion into the common bile duct for in situ cold plasma delivery.
Analytical techniques
In addition to the analytical methods used by our biological partners—such as RNA sequencing, flow cytometry, ultra-microscopy, and access to animal facilities and research platforms—the LPP has shared equipment enabling detailed characterization of the plasma phase and its interaction with various biological targets:
Electrical diagnostics: current probes, high-voltage probes, electric field probes
Spectroscopy techniques: optical emission spectroscopy, mass spectrometry, Fourier-transform infrared spectroscopy (FTIR)
Imaging techniques: infrared thermographic imaging, ultra-fast ICCD imaging





